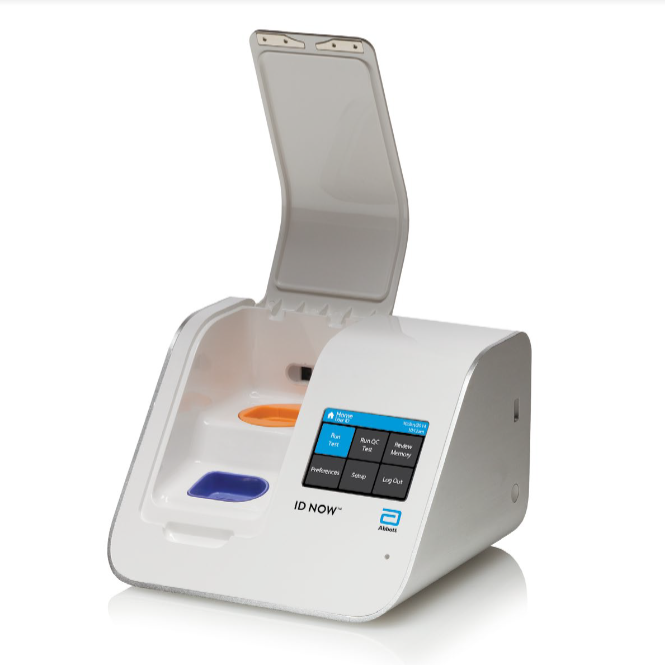
COVID-19 Rapid testing
Rapid test for identification of COVID-19 virus
Results are for the identification of SARS-CoV-2 RNA, demonstrating virus presence during active virus infection.
ID NOW™ COVID-19 assay performed on the ID NOW™ Instrument is a rapid molecular in vitro diagnostic test utilizing an isothermal nucleic acid amplification technology intended for the qualitative detection of nucleic acid from the SARS-CoV-2 viral RNA in direct nasal, nasopharyngeal or throat swabs from individuals who are suspected of COVID-19 by their healthcare provider. The ID NOW™ COVID-19 assay is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver.
ID NOW™ COVID-19 is a rapid (13 minutes or less), instrument-based isothermal test for the qualitative detection and diagnosis of SARS-CoV-2 from nasal, nasopharyngeal and throat swabs